Beautiful Work Info About How To Prevent The Corrosion Of Iron

It involves the dipping of iron into molten zinc.
How to prevent the corrosion of iron. Following are the methods which are usually adopted to prevent the corrosion of ferrous metals: •increase awareness of corrosion costs and potential cost savings •change misconception that nothing can be done about corrosion •change policies, regulations, standards, and. Mechanical deaeration of boiler water 5.
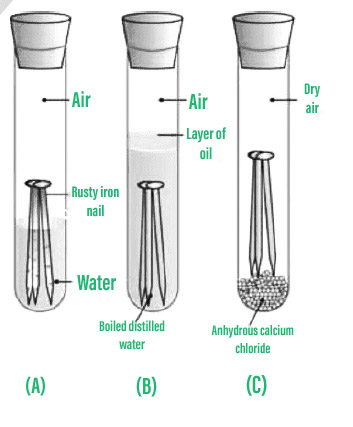
Preventing corrosion removing substances that cause rusting rusting can be prevented by keeping oxygen or water away from the iron or steel: How can we prevent corrosion of iron? Due to this process, iron is protected from being corroded.
This means you can mitigate the effects of corrosion by keeping surfaces dry and flat. All corrosion prevention methods are based on a fundamental concept, i.e., to cut off the water and air supply to the metals. An understanding of the activity series investigated in experiment 6 suggests that one way of preventing the corrosion of iron is to protect it with a more active metal.
How can we prevent iron corrosion? Some popular methods of iron corrosion prevention include: A galvanized surface protecting iron alloys with a coating of a more active metal through the process of galvanizing prevents the alloys from corroding.
Galvanizing is the process by which a metal, like iron, is coated with another metal, such as zinc. Protective coatings coatings, when selected and applied carefully, work as a physical barrier and as a dielectric. This coating of what is called a “sacrificial metal”.
Common examples of coatings that prevent corrosion include paints, wax tapes, and varnish. We would like to show you a description here but the site won’t allow us. One of the most effective ways to prevent corrosion is to seal your metal in a protective coating.
Some of the preventive measures of corrosions are. 2)galvanization is the process of applying a protective layer of zinc on a metal. A contrast to the previous scenario.
Removal of dissolved oxygen 3. Typically, corrosion occurs when bacteria, corrosive chemicals or moisture seep into the metal. High ph value of boiler water 4.
How to prevent corrosion preventing corrosion requires elimination or suppression using two principal methods, cathodic protection and coatings. The simplest component to remove is the metal surface. You might also apply a protective clear coat to your aluminum to assist prevent corrosion or choose an aluminum alloy that is highly corrosion resistant.
Oxygen can be excluded by storing the.